[Hygiène prévention Covid-19 (French)] - [Pourquoi ces survies différentes du Covid sur plastique et papier ?] - [Why Does Sars-Cov-2 Survive Longer on Plastic Than on Paper?, Medical Hypotheses
Why is coronavirus inactivated faster on paper than on plastic surface?
What are the mechanisms of survival and destruction of virus on solid surfaces?
August 3, 2020
 Why does this Covid-19 coronavirus survive a thousand times longer on mask & plastic than on cotton, paper and tissue?
Why does this Covid-19 coronavirus survive a thousand times longer on mask & plastic than on cotton, paper and tissue?
Covid-19 coronavirus survives*1 much longer on smooth surfaces (e.g. glass and plastic: up to 7 days) than on paper (less than 3 hours). By classifying materials according to virus stability of the on their surface we obtain the following list (from long to short stability):
polypropylene (mask), plastic, stainless steel, glass, skin*2, cardboard, wood, cotton, copper, paper, tissue
(Chin&Poon_Lancet.Micr.20 ; Van-Doremalen_NEJM*2020 ; Harbourt_Rx* ).
This leads us to a pragmatic conclusion and to a scientific question:
- Pragmatically, these differences suggest we should use paper instead of plastic to wrap or cover objects (books, food): this would reduce the spread of virus.
- Scientifically, these differences in survival time are puzzling: Why would the virus be "killed" by certain materials, and conversely be "protected" by others?
Intuitively, I have the childish feeling that absorbent products, “blotters”, can kill the virus. In contrast, the smooth and waterproof materials seem to protect the virus (except copper). Would the virus be killed by the lack of water? How can we accept such a simple explanation? A virus is not really alive, and it.does not seem to contain water: How could a virus die of "dryness"?
However, it seems that some viruses could be inactivated by desiccation. Cox stated 30 years ago that when water disappears from the environment of a microorganism with a double layer membrane (as bacteria and enveloped viruses), it quickly loses its viability by oxidation and Maillard reactions (Cox_OLEB_1993). The fat envelope would hold only with water on both sides of its two lipidic layers. This was repeated in recent articles without any experimental evidence (Casanova-AEM-2010 ; Castaño_Rx*20).
Moreover, a detrimental effect of surface porosity is found on the survival of other enveloped viruses:
- The influenza virus survives 24-48h on plastic and stainless steel, but less than 8h on paper and tissue (often only a few minutes). The authors suggest that porous surfaces capture moisture and consequently alter the viability of influenza virus (Bean&Balfour_JID_1982).
- Common cold coronaviruses survive less than an hour when they are dried up (Vasickova_FEV_2010)
In addition, the belief that viruses do not contain water was proven false: In 1950 Sharp & Beard established by clever experiments on influenza viruses (influenza A, B and swine) that these viruses contain about 60% water (between 150 and 230% of their dry weight, according to strain and method). These viruses are "enveloped" in a lipid membrane, just like coronavirus (Sharp&Beard_JBC_1950 )
Finally, water droplets can stay for days on plastic: Kumar & Hong demonstrate, through photos taken under a microscope every hour, that drops first shrink rapidly on a smooth surface. Then, the resulting microdrops persist for more than 24 hours on plastic, without further decrease in size. They stay less on stainless steel, and they disappear from a copper surface. These microdrops are 10 to 100 times larger than a coronavirus, which is enough to shelter the virus.
Kumar & Hong explain disappearance of drops on copper surface by the high copper thermal conductivity: metal heat would lead to fast evaporation of droplets, while drops would be protected from heat on more insulating surfaces like stainless steel and plastic.
Finally Kumar & Hong show that 95% of these droplets can be removed from a plastic surface by wiping it with a tissue. Sadly, the authors did not study the disappearance of drops on paper or cotton, but we can assume that they quickly disappear from these absorbent supports, leaving viruses exposed to dry air unprotected, thus leading to their fast inactivation. (Kumar&Hong_Rx*20)
Copper is an outlier in the above cited list of materials: it is the only smooth and waterproof surface on which the virus persists less than a day. This can be explained, in addition to Kumar & Hong's speculation, by its antiseptics properties linked to the pro-oxidant activity of Cu++ ions. (Noyce&Keevil_AEM_2007 ; Warnes&Keevil_mBio_2015). Silver has not yet been tested with coronavirus. Like copper, it has a high thermal conductivity and is also a powerful antiseptic. We therefore assume that coronavirus would not survive longer on silver than on copper.
Finally, on protective masks, coronavirus persists for a particularly long time.
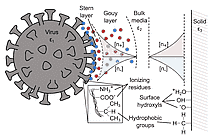 The mask inner layer (N ° 2) is made of a specific plastic material, polypropylene, which holds permanent electrostatic charges: it is thus called an "electret" (Heaviside_The Electrician_1885). The stability of viruses on this material may be linked to the strong bond between polypropylene electret and the viral particles (See Fig.3 in Castaño_Rx*20) . Virus adsorption on surface stabilize them (Vasickova_FEV_2010), for instance, survival of viruses in soil is a direct function of their adsorption onto soil particles (Hurst-AEM-1980). This was not, however, demonstrated for coronavirus on mask electret.
The mask inner layer (N ° 2) is made of a specific plastic material, polypropylene, which holds permanent electrostatic charges: it is thus called an "electret" (Heaviside_The Electrician_1885). The stability of viruses on this material may be linked to the strong bond between polypropylene electret and the viral particles (See Fig.3 in Castaño_Rx*20) . Virus adsorption on surface stabilize them (Vasickova_FEV_2010), for instance, survival of viruses in soil is a direct function of their adsorption onto soil particles (Hurst-AEM-1980). This was not, however, demonstrated for coronavirus on mask electret.
In conclusion, we suggest that dryness kills SARS-CoV-2 viruses on paper and cotton. Conversely, the droplets of water that stay for days on a plastic surface would protect the viruses. The formal demonstration of this hypothesis remains however to be done experimentally with paper.
- This demonstration would be of scientific interest, explaining the survival mechanisms of lipid bilayer enveloped viruses on solid surfaces.
- It would also make it possible to predict whether the enveloped viruses may persist on materials that have not been tested yet, thereby reducing the transmission of viruses by objects, and therefore the spread of the pandemic *3.
Notes
*1 In the above text, the term "survival" applied to the coronavirus is a misnomer, as well as all those who evoke the virus life. In fact, viruses are not "alive" in the ordinary meaning of the term, since they must be internalized in a living cell to reproduce.
*2 Survival of the Covid19 virus on human skin has not yet been studied, but only on a piece of pig skin, obtained post-mortem (Harbourt_Rx*)
*3 The hypothesis predicts for example that these coronaviruses, and all enveloped viruses will be quickly inactivated on absorbent products like newspaper, leather returned "suede", chalk or salt. Conversely viruses will be relatively stable on poorly absorbent products like glossy paper, polished marble or fruit skin, and evne more stable on wet products like meat fresh.
My E-mail is shown at bottom of page Denis
 Why does this Covid-19 coronavirus survive a thousand times longer on mask & plastic than on cotton, paper and tissue?
Why does this Covid-19 coronavirus survive a thousand times longer on mask & plastic than on cotton, paper and tissue? The mask inner layer (N ° 2) is made of a specific plastic material, polypropylene, which holds permanent electrostatic charges: it is thus called an "electret" (Heaviside_The Electrician_1885). The stability of viruses on this material may be linked to the strong bond between polypropylene electret and the viral particles (See Fig.3 in Castaño_Rx*20) . Virus adsorption on surface stabilize them (Vasickova_FEV_2010), for instance, survival of viruses in soil is a direct function of their adsorption onto soil particles (Hurst-AEM-1980). This was not, however, demonstrated for coronavirus on mask electret.
The mask inner layer (N ° 2) is made of a specific plastic material, polypropylene, which holds permanent electrostatic charges: it is thus called an "electret" (Heaviside_The Electrician_1885). The stability of viruses on this material may be linked to the strong bond between polypropylene electret and the viral particles (See Fig.3 in Castaño_Rx*20) . Virus adsorption on surface stabilize them (Vasickova_FEV_2010), for instance, survival of viruses in soil is a direct function of their adsorption onto soil particles (Hurst-AEM-1980). This was not, however, demonstrated for coronavirus on mask electret.